Bi-Mettalic Reactions
Introduction
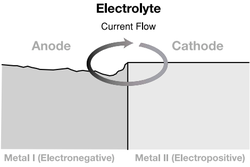
Bi-metallic reactions are a natural phenomenon which must be given due consideration when designing your facade. Bi-metallic reaction is the corrosive effect which occurs when 2 dissimilar metals are connected or in contact with each other.
The reaction occurs when:
Materials
The corrosion risks of dis-similar materials can be simply quantified as:
> Additional corrosion unlikely
> Additional corrosion may occur
> Additional corrosion possible
The galvanic chart places metals in order of anodic to cathodic, with the net result being that the greater the distance the metals are from each other on the chart, the greater the risk of reaction. The reaction is that the cathodic metal attacks the anodic metal.
Galvanic Chart:
ANODIC END
For the purpose of normal facade materials, the reactions to be aware of and to be prepared to compensate for would be defined as follows:
> Galvanised steel frames (Zinc)
> Aluminium Extrusions
> Aluminium sheets
> 304 stainless steel
> 316 stainless steel
> Copper (electrical continuity tapes)
Procedures
With the exception of Copper, typically the metals are close on the galvanic chart and as such can be considered a low risk depending on the environmental conditions.
Methods of preventing bi-metallic corrosion are simply to break the electrical path or omit the accelerating factors of oxygen or water depending on the type of connection being considered.
Electrical insulation can be achieved with the use of isolators, such as nylon, PTFE, EPDM etc. or by painting or applying sacrificial coatings (eg Zinc).
For example, you would not consider the effect between zinc and aluminium to be hazardous when within a controlled internal environment. However, when external you would need to give due consideration, but this can be done simply by either isolating the contact between the metals, or sometimes more simply, by covering them to protect them from the external environmental conditions of moisture or humidity.
For copper, especially as this is used for electrical continuity connections, this is typically done by considering the galvanic chart, whereby stainless steel is close to copper, and also to aluminium, hence you can use the stainless steel as the isolator, i.e.: copper tape - Stainless steel washer - aluminium frame/bracket, whereby the stainless steel maintains the electrical continuity whilst at the same time providing the metallic isolation.
Other considerations need to be given to the size/area of contact and materials. For example, a small Cathode in contact with a large Anode (i.e. stainless steel fixings in aluminium), will give insignificant reaction. However the reverse condition could be the opposite, with significant reaction possible. Accordingly it could be considered a risk to use aluminium rivets to fix stainless steel sheets, as the rivet would (as the Anode) be at risk of corrosion.
References
PD6484: Commentary on Corrosion at Bi-metallic contacts
NPL Guide to good practice in corrosion control
Bi-metallic reactions are a natural phenomenon which must be given due consideration when designing your facade. Bi-metallic reaction is the corrosive effect which occurs when 2 dissimilar metals are connected or in contact with each other.
The reaction occurs when:
- An electrolyte bridges two metals
- Electrical continuity is achieved between two metals
- A sufficient difference in potential is achieved between two metals
- A sustained cathodic reaction on one of the metals occurs
Materials
The corrosion risks of dis-similar materials can be simply quantified as:
> Additional corrosion unlikely
> Additional corrosion may occur
> Additional corrosion possible
The galvanic chart places metals in order of anodic to cathodic, with the net result being that the greater the distance the metals are from each other on the chart, the greater the risk of reaction. The reaction is that the cathodic metal attacks the anodic metal.
Galvanic Chart:
ANODIC END
- Magnesium
- Magnesium alloys
- Zinc
- Aluminium grades 5052, 3004, 3003, 1100, 6053
- Cadmium
- Aluminium grades 2117, 2017, 2024
- Mild steel (1018), wrought iron
- Cast iron, low alloy high strength steel
- Chrome iron (active)
- Stainless steel, 430 series (active)
- Stainless steel grade 302,303,304,321,347,410,416 (active)
- NI - Resist
- Stainless steel grade 316, 317 (active)
- Carpenter 20 CB-3 Stainless (active)
- Aluminium bronze (CA687)
- Hastelloy (active), Inconel (active, Titanium (active)
- Lead - Tin solders
- Lead
- Tin
- Inconel 600 (active)
- Nickel (active)
- 60 NI-15 CR (active)
- 80 NI-20 CR (active)
- Hastelloy B (Active)
- Brasses
- Copper
- Manganese Bronze (CA675), Tin Bronze (CA903, 905)
- Silicon Bronze
- Nickel Silver
- Copper-Nickel alloy 90-10
- Copper-Nickel alloy 80-20
- Stainless steel grade 430
- Nickel, Aluminium, Bronze (CA630,632)
- Monel 400, K500
- Silver Solder
- Nickel (passive)
- 60 NI -15 CR (passive)
- Inconel 600 (passive)
- 80 NI - 20 CR (passive)
- Chrome Iron (passive)
- Stainless steel grade 302,303,304,321,347 (passive)
- Stainless steel grade 316,317 (passive)
- Carpenter 20 CB-3 Stainless (passive), Incoloy 825
- Nickel - Molybdeum - Chromium - Iron alloy (passive)
- Silver
- Titanium (passive), Hastelloy C & C276 (passive, Inconel 625 (passive)
- Graphite
- Zirconium
- Gold
- Platinum
For the purpose of normal facade materials, the reactions to be aware of and to be prepared to compensate for would be defined as follows:
> Galvanised steel frames (Zinc)
> Aluminium Extrusions
> Aluminium sheets
> 304 stainless steel
> 316 stainless steel
> Copper (electrical continuity tapes)
Procedures
With the exception of Copper, typically the metals are close on the galvanic chart and as such can be considered a low risk depending on the environmental conditions.
Methods of preventing bi-metallic corrosion are simply to break the electrical path or omit the accelerating factors of oxygen or water depending on the type of connection being considered.
Electrical insulation can be achieved with the use of isolators, such as nylon, PTFE, EPDM etc. or by painting or applying sacrificial coatings (eg Zinc).
For example, you would not consider the effect between zinc and aluminium to be hazardous when within a controlled internal environment. However, when external you would need to give due consideration, but this can be done simply by either isolating the contact between the metals, or sometimes more simply, by covering them to protect them from the external environmental conditions of moisture or humidity.
For copper, especially as this is used for electrical continuity connections, this is typically done by considering the galvanic chart, whereby stainless steel is close to copper, and also to aluminium, hence you can use the stainless steel as the isolator, i.e.: copper tape - Stainless steel washer - aluminium frame/bracket, whereby the stainless steel maintains the electrical continuity whilst at the same time providing the metallic isolation.
Other considerations need to be given to the size/area of contact and materials. For example, a small Cathode in contact with a large Anode (i.e. stainless steel fixings in aluminium), will give insignificant reaction. However the reverse condition could be the opposite, with significant reaction possible. Accordingly it could be considered a risk to use aluminium rivets to fix stainless steel sheets, as the rivet would (as the Anode) be at risk of corrosion.
References
PD6484: Commentary on Corrosion at Bi-metallic contacts
NPL Guide to good practice in corrosion control
